Laboratory of the Sliding Koala
Recent Publications
Controlling Gas Selectivity by Tuning the Molecular Porous Liquids
|
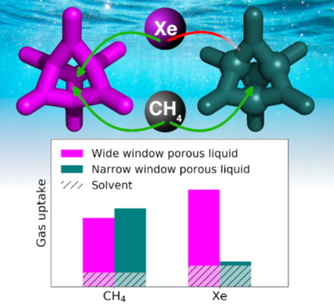
Controlling Gas Selectivity in Molecular Porous Liquids by Tuning the Cage Window Size
Benjamin D. Egleston, Konstantin V. Luzyanin, Michael C. Brand, Rob Clowes, Michael E. Briggs, Rebecca L. Greenaway, and Andrew I. Cooper Control of pore window size is the standard approach for tuning gas selectivity in porous solids. Here, we present the first example where this is translated into a molecular porous liquid formed from organic cage molecules. Reduction of the cage window size by chemical synthesis switches the selectivity from Xe‐selective to CH4‐selective, which is understood using 129Xe, 1H, and pulsed‐field gradient NMR spectroscopy. Angew. Chem. Int. Ed. 2020, 59, 7362–7366. DOI: 10.1002/anie.201914037 https://doi.org/10.1002/anie.201914037 |
Impact of Halogen Bonding on Solubility of Organometallics
|
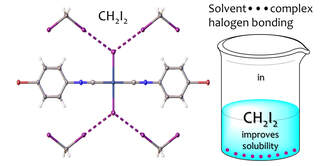
Dramatically Enhanced Solubility of Halide‐Containing Organometallic Species in Diiodomethane: The Role of Solvent⋅⋅⋅Complex Halogen Bonding
Mikhail A. Kinzhalov, Mariya V. Kashina, Alexander S. Mikherdov, Ekaterina A. Mozheeva, Alexander S. Novikov, Andrey S. Smirnov, Daniil M. Ivanov, Mariya A. Kryukova, Aleksandr Yu. Ivanov, Sergej N. Smirnov, Vadim Yu. Kukushkin, and Konstantin V. Luzyanin Exceptional solvation properties of diiodomethane and its inertness towards organometallics make this solvent a good candidate for NMR solvent-of-choice, in particular, for the acquisition of insensitive spins. Better solvation properties of diiodomethane were associated with formation of the uniquely strong solvent-(metal complex) halogen bonding. Angew. Chem. Int. Ed. 2018, 130, 12967–12971. DOI: 10.1002/ange.201807642 https://doi.org/10.1002/ange.201807642 |
Visible Light Photocatalysis using Self-Photosensitising Catalysts
|
Visible light accelerated hydrosilylation of alkynes using platinum–[acyclic diaminocarbene] photocatalysts
Jack C. Gee, Beth A. Fuller, Hannah-Marie Lockett, Gita Sedghi, Craig M. Robertson, and Konstantin V. Luzyanin Platinum–[diaminocarbene] complexes work as transition-metal photocatalysts for the hydrosilylation of alkynes under blue light irradiation. Chem. Commun., 2018, 9450–9453. DOI: 10.1039/C8CC04287J http://dx.doi.org/10.1039/C8CC04287J |
(C) Dr. Konstantin Luzyanin, 2021.